- Blog
- Ready no backup exec servers are available
- Chilean mauser caliber
- Fabfilter total bundle torrent
- Dr paul dobransky women
- Schrodinger atomic model
- Speedconnect internet accelerator v8-0 download
- Xbox 360 s amazon
- Halo 2 pc vs xbox
- Cyberlink youcam 7 deluxe download
- Buy far cry 4 pc
- Overlord raising hell g2a
- Printopia torrent
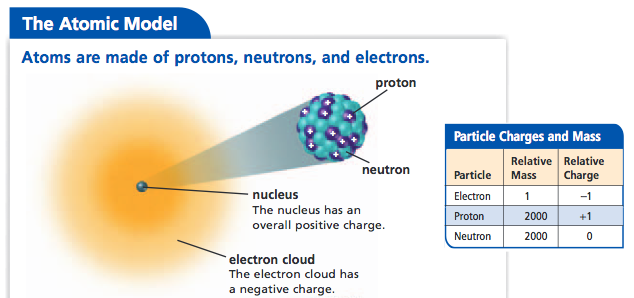
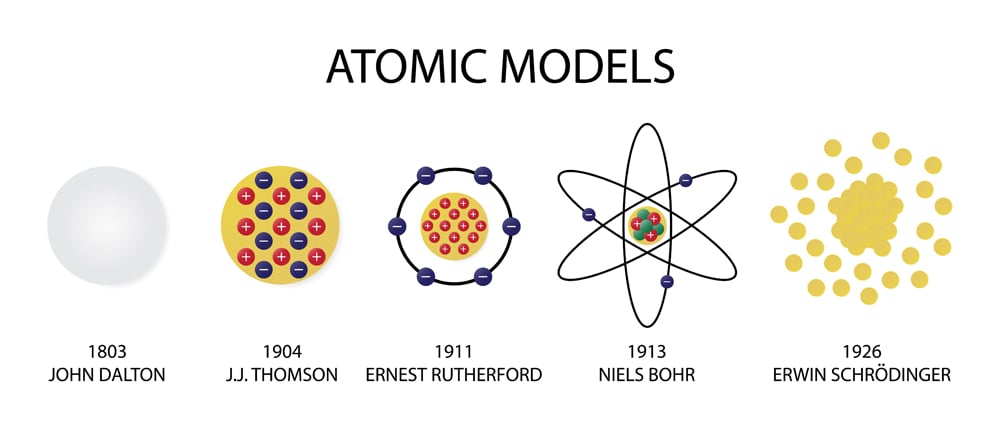
He tried to reconcile the idea of life as "pure mechanism" with our experience of ourselves as individuals directing our own actions. In a curious epilogue, Schrödinger revealed that his interests were not exclusively scientific. Schrödinger's little book had an enormous impact both James Watson and Francis Crick separately claimed it as a major influence on their decision to pursue the structure of DNA. He saw life as a finely tuned mechanism, "the finest masterpiece ever achieved along the lines of the Lord's quantum mechanics." Schrödinger realized that the latter process is not dissimilar to what we see in a clock or any other mechanical object. (heavy protons, light electrons) Classical planetary motion was solved by Newton. He based this model on the results of Geiger and Marsden for scattered particles, which showed that most of the mass of an atom was concentrated at the nucleus. There are two ways of producing order, according to Schrödinger, 'order from disorder' as we see in the statistical description of large numbers of atoms, or 'order from order,' which seems to be the way that life works. Rutherford-Bohr atom In 1912 Ernest Rutherford proposed a planetary model of the hydrogen atom. He imagined the chromosomes to be a kind of extraordinarily stable irregular crystal (a model he borrowed from Max Delbrück), "an unusually large molecule, which has to be a masterpiece of differentiated order." The atoms within the 'chromosome molecule' were arranged to produce a 'code script' that Schrodinger explicitly compared to Morse code. seems to be connected with the presence of the 'aperiodic solids', the chromosome molecules." Schrödinger never mentions DNA, which had yet to be confirmed as the source of heredity.
"An organism's astonishing gift of concentrating a 'stream of order' on itself and thus escaping the decay into atomic chaos. "What an organism feeds upon is negative entropy." Metabolism, for him, was the export of entropy to the surrounding environment. "It is by avoiding the rapid decay into the inert state of 'equilibrium' that an organism appears so enigmatic," wrote Schrödinger, in a small monograph titled What is Life? in 1945.

A classical physicist trying to understand biology, according to Schrödinger, was like an early mechanic, ignorant of electricity, trying to understand the principles behind an electric motor. But he also turned his physicists' intuition to the problem of biology.įor a physicist of the 1940s, life was indeed a conundrum it seemed to violate all the statistical methods with which they were used to describing physical systems. Erwin Schrödinger won the Nobel Prize for his famous equation that describes the changes in a quantum system over time, and he is probably even better known for his poor quantum cat, which was simultaneously alive and dead.
- Blog
- Ready no backup exec servers are available
- Chilean mauser caliber
- Fabfilter total bundle torrent
- Dr paul dobransky women
- Schrodinger atomic model
- Speedconnect internet accelerator v8-0 download
- Xbox 360 s amazon
- Halo 2 pc vs xbox
- Cyberlink youcam 7 deluxe download
- Buy far cry 4 pc
- Overlord raising hell g2a
- Printopia torrent
